 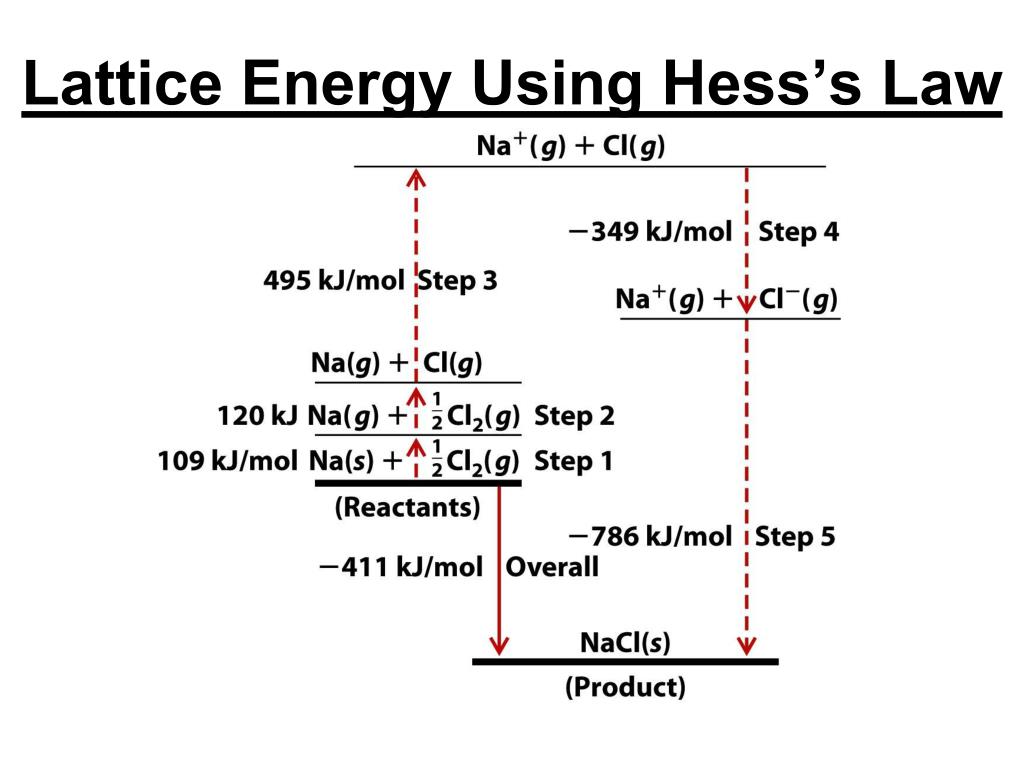
Lattice Energy Examples When sodium ion (Na+) combines with chloride ion (Cl–), sodium chloride (NaCl) crystal forms, and 787.3 kJ of energy is released. The bond between ions of opposite charge is strongest when the ions are small. The lattice energy of NaCl, for example, is 787.3 kJ/mol, which is only slightly less than the energy given off when natural gas burns. ∆H4 = Electron affinity of Cl(s) = – 349 kJ mol-1.∆H3 = dissociation energy of Cl2(s) = 244 kJ mol-1.∆H2 = ionisation energy of Na(s) = 495 kJ mol-1.∆H1 = heat of sublimation of Na(s) = 108.7 kJ mol-1.∆Hf = heat of formation of sodium chloride = 411.3 kJ mol-1.How do you find the lattice energy of NaCl? Lattice energies cannot be measured directly but are obtained from a thermochemical cycle called the Born–Haber cycle, in which Hess’s law is used to calculate the lattice energy from the measured enthalpy of formation of the ionic compound, along with other thermochemical data. How do you find the lattice energy of a crystal? Instead, the lattice energy is calculated by subtracting the other four energies in the Born–Haber cycle from the net enthalpy of formation. The net enthalpy of formation and the first four of the five energies can be determined experimentally, but the lattice energy cannot be measured directly. How do you find the lattice energy of a Born-Haber cycle? This Lattice Energy Formula is as follows: U=−rack′Q_1Q_2r_0 U is always a positive number, and it represents the amount of energy required to dissociate 1 mol of an ionic solid into the gaseous ions. We can compute the lattice energy of nearly any ionic solid by using a modified form of Coulomb’s law. There are several different equations, of various degrees of complication, for calculating lattice energy in this way.24 Which has higher lattice energy NaCl or MgO? What is lattice energy calculate lattice energy? If you know how to do it, you can then fairly easily convert between the two. Calculations of this sort end up with values of lattice energy, and not lattice enthalpy. By doing physics-style calculations, it is possible to calculate a theoretical value for what you would expect the lattice energy to be. Let's also assume that the ions are point charges - in other words that the charge is concentrated at the center of the ion. Let's assume that a compound is fully ionic. Theoretical Estimates of Lattice Energies Once again, the cycle sorts out the sign of the lattice enthalpy. This time both routes would start from the elements in their standard states, and finish at the gaseous ions. You cannot use the original one, because that would go against the flow of the lattice enthalpy arrow. It does, of course, mean that you have to find two new routes. The only difference in the diagram is the direction the lattice enthalpy arrow is pointing. How would this be different if you had drawn a lattice dissociation enthalpy in your diagram? Your diagram would now look like this: So, from the cycle we get the calculations directly underneath it. The diagram is set up to provide two different routes between the thick lines. Now we can use Hess' Law and find two different routes around the diagram which we can equate. And finally, we have the positive and negative gaseous ions that we can convert into the solid sodium chloride using the lattice formation enthalpy.Remember that first electron affinities go from gaseous atoms to gaseous singly charged negative ions. 
The -349 is the first electron affinity of chlorine.Again, we have to produce gaseous atoms so that we can use the next stage in the cycle. The +122 is the atomization enthalpy of chlorine.Remember that first ionization energies go from gaseous atoms to gaseous singly charged positive ions. The +496 is the first ionization energy of sodium.We have to produce gaseous atoms so that we can use the next stage in the cycle. The +107 is the atomization enthalpy of sodium.The Born-Haber cycle now imagines this formation of sodium chloride as happening in a whole set of small changes, most of which we know the enthalpy changes for - except, of course, for the lattice enthalpy that we want to calculate. The arrow pointing down from this to the lower thick line represents the enthalpy change of formation of sodium chloride. Notice that we only need half a mole of chlorine gas in order to end up with 1 mole of NaCl. 
We are starting here with the elements sodium and chlorine in their standard states. If you wanted to draw it for lattice dissociation enthalpy, the red arrow would be reversed - pointing upwards.įocus to start with on the higher of the two thicker horizontal lines. You will see that I have arbitrarily decided to draw this for lattice formation enthalpy. \)Ĭonsider a Born-Haber cycle for sodium chloride, and then talk it through carefully afterwards. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
